This post is a continuation of a series I started a few months ago, looking at old and new scenarios for the early evolution of animals. This thinking was prompted by a pair of of conferences I went to over the last few years, one in Tübingen and one in London.
The first animals evolved in the sea. That setting brings with it many consequences – a particular chemical environment, and physical features such as ease of motion. The first animals may have floated about as semi-organized balls of cells. The sea is a truly three-dimensional world. Some time after the earliest days, an important event was the invention of bilaterally symmetrical animals – or bilaterians – those with a left and right as well as a top and bottom. We are bilaterians, while jellyfish and sponges are not. Much stems from our bilaterial symmetry – our forms of motion, the ways our sensing and behavior are organized. This is so familiar a layout for animals that it takes a while to get used to the fact that animals can be laid out differently. How did bilateral symmetry evolve? One family of possibilities involve life on the sea floor, and how animals came to make a living in that more two-dimensional, land-like milieu.
The animals in this post are nudibranchs in the Armina genus. I am not completely sure of the species, and this is one of the groups that’s still being sorted out. The one above might be Armina cygnea – the swan-like Armina. I saw them for the first time earlier this year, cruising the sea floor at Jervis Bay in a stately bilateral way.
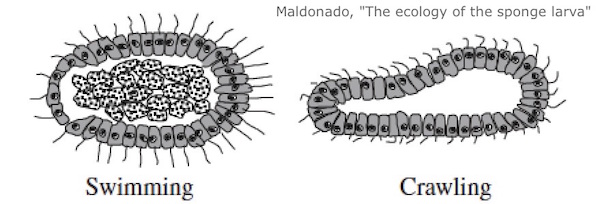
A while ago I wrote about an evolutionary scenario due to Claus Nielsen. Nielsen is guided by the idea that sponges are a plausible early form of animal life. The fact that sponges are so far from us, and even the fact that their bodies are simple, does not imply that they are an “ancestral” form. But other lines of evidence suggests they might be. Nielsen’s paper was subtitled “Are we derived sponge larvae?” His scenario works like this. 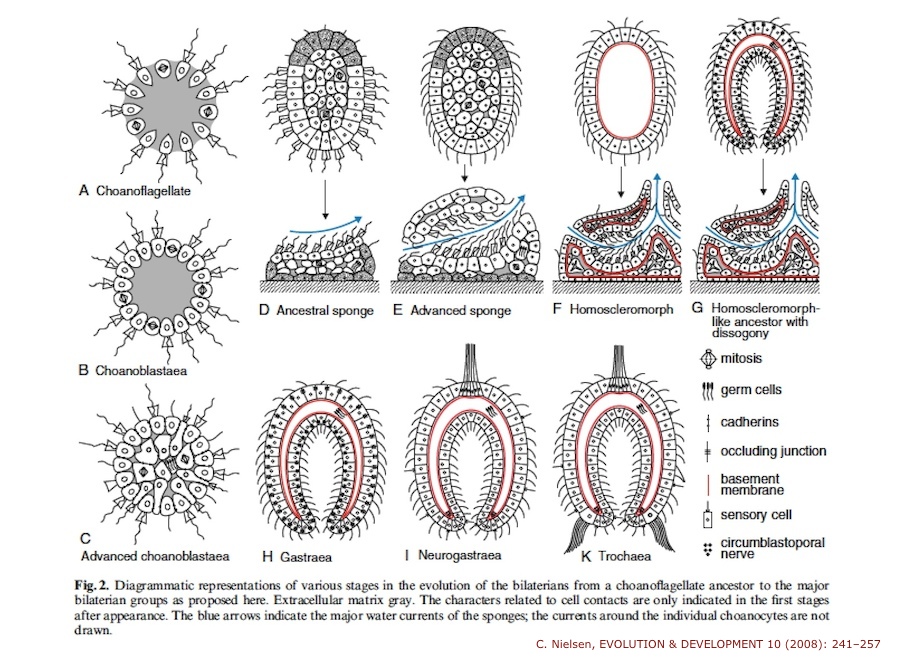 Sponge adults never (or almost never) move, but their lavae, the juvenile forms, can swim. Nielsen sees the swimming sponge larva as a plausible precursor of other animals. This would involve a process in which a juvenile form of an animal becomes reproductively mature. Some larvae keep swimming, reproduce, and give rise to other animals, eventually including bilaterians. In my talk at the Tübingen conference, I said that sponge larvae might have been important through a different path. Sponge larvae have no nervous systems, but do have various sensors, and they swim with cilia, little hairs. They swim around, sense a suitable place to settle, and then anchor themselves and transform. We can then imagine a scenario in which a larva who departs from the usual sponge life cycle does not remain in the water column, but lands on the sea floor. What I’m imagining is a failed metamorphosis; the larva uses its sensory apparatus to find a place to settle, but metamorphosis fails to occur. The result is a departure from the sponge life cycle that produces a crawling form.
Sponge adults never (or almost never) move, but their lavae, the juvenile forms, can swim. Nielsen sees the swimming sponge larva as a plausible precursor of other animals. This would involve a process in which a juvenile form of an animal becomes reproductively mature. Some larvae keep swimming, reproduce, and give rise to other animals, eventually including bilaterians. In my talk at the Tübingen conference, I said that sponge larvae might have been important through a different path. Sponge larvae have no nervous systems, but do have various sensors, and they swim with cilia, little hairs. They swim around, sense a suitable place to settle, and then anchor themselves and transform. We can then imagine a scenario in which a larva who departs from the usual sponge life cycle does not remain in the water column, but lands on the sea floor. What I’m imagining is a failed metamorphosis; the larva uses its sensory apparatus to find a place to settle, but metamorphosis fails to occur. The result is a departure from the sponge life cycle that produces a crawling form.
One thing that makes me think this is worth considering is the fact that some present-day sponge larvae do crawl.
Once an animal is crawling, a quasi-bilaterian organization may be easy to acquire. The top and bottom of the animal have natural roles, as well as the pre-existing front-versus-back distinction. A partly bilaterian form might come and go in evolution more easily than often supposed. We can imagine a process in which a partly bilaterian form is produced by a flexible or “plastic” response of the organism to contact with the sea floor. Over evolutionary time, this response becomes “canalized” or genetically assimilated – it becomes a routine part of the organism’s development.
A scenario like this connects in interesting ways to the fossil record and our picture of ancient environments. The first animal fossils are from the Ediacaran, around 600 million years ago. It is thought that in the Ediacaran there were rich microbial mats on the sea floor, and at least some animals made a living there, existing (in the paleontologist Adolf Seilacher’s words) as “mat encrusters, mat scratchers, mat stickers, and undermat miners.”
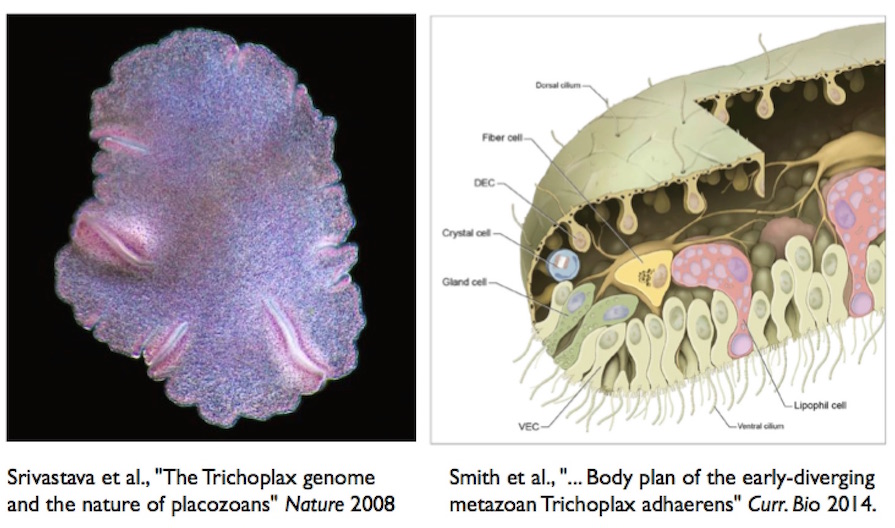
Nielsen’s scenario supposes that the sponge larva had evolved a gut before it abandoned the formerly adult stage. Suppose instead that such a larva began a crawling life before it had a gut. The result might come to look like a present-day placozoan. These animals have no gut, and crawl on a surface with cilia, digesting food below them externally. Their bodies are not symmetrical at all.
The current placement of placozoans on the phylogenetic tree has them diverging from bilaterians more recently than sponges, earlier than cnidarians (jellyfish, corals, etc). Suppose a sponge larva adopted a crawling life, and diversified. One road leads to a gut and a greater degree of organization, including a nervous system; another road might lead to a simple flattening, which maximizes the area available for external digestion, and a placozoan sort of life.
So I wonder whether a sponge larva might have adopted a sea-floor crawling life, a bit like a very simple slug, and diversified into a number of animals feeding on the microbial mats.
This picture is a relative of a much earlier one, the bilaterogastraea hypothesis developed by the Swede Gösta Jägersten in the mid 20th century. Jägersten, as I understand it, did not take us through sponges, but thought that animal evolution began when a ball of cells – a blastula – descended to the sea floor, became bilaterally organized, and acquired a gut (hence bilaterogastraea). This view seems not to have been taken very seriously, as there was not much evidence for it. The picture sketched above is a modification of the Nielsen view, which is based on much evidence unavailable to Jägersten, but it has some similarities. I’ll continue this theme in some later posts, looking at other options. All this material is speculative, but one never knows which hypotheses will turn out – so to speak – to have legs.
________________
Notes:
Sperling and Vinther (2010) have argued that the Ediacaran organism Dickinsonia might have had a placazoan affinity. Dickinsonia seems to have been a flat, bottom-dwelling animal which settled, moved, and settled again. It was much larger than the tiny present day placazoans, and more complex – it is sometimes seen as bilaterian. But it might not have had a gut and it may have fed, Sperling and Vinther think, in the same way placazoans do, by absorbing nutrients from its bottom surface.
About my title.
A couple of additional nudibranch seafloor explorers have been posted at the Instagram site that now accompanies this one.